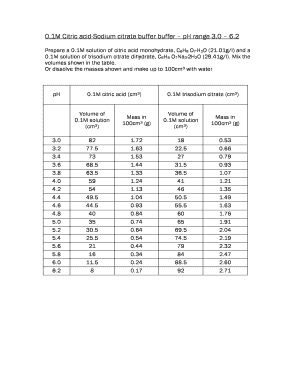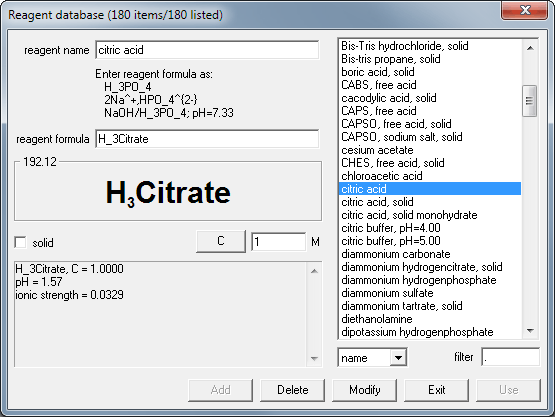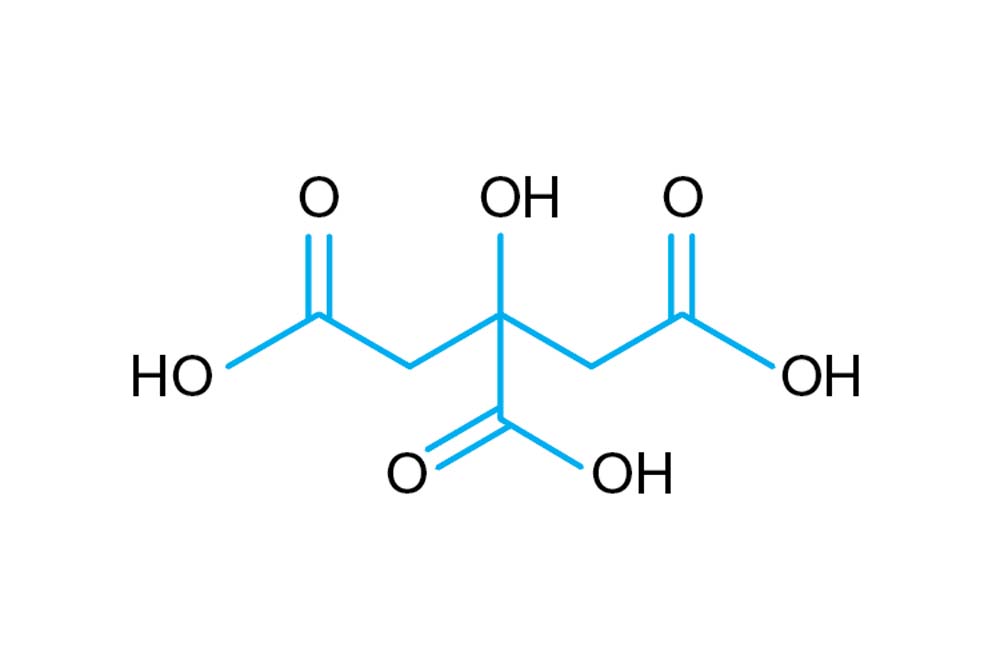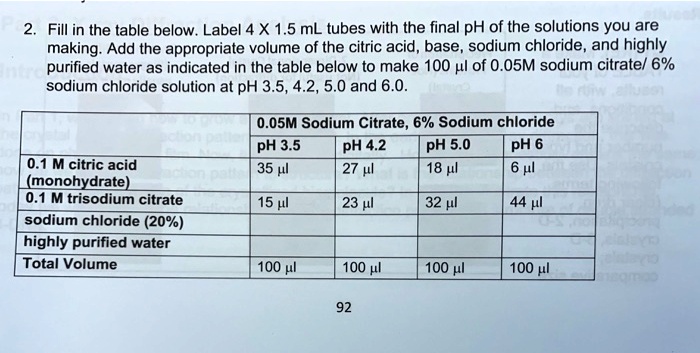
SOLVED: Text: Fill in the table below. Label 4 X 1.5 mL tubes with the final pH of the solutions you are making. Add the appropriate volume of the citric acid, base,
![pH 4.25 Sodium Citrate Buffer Solution・199-07185[Detail Information] | [Analytical Chemistry]|Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation pH 4.25 Sodium Citrate Buffer Solution・199-07185[Detail Information] | [Analytical Chemistry]|Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation](https://labchem-wako.fujifilm.com/sc/06/199-07185.jpg)
pH 4.25 Sodium Citrate Buffer Solution・199-07185[Detail Information] | [Analytical Chemistry]|Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation
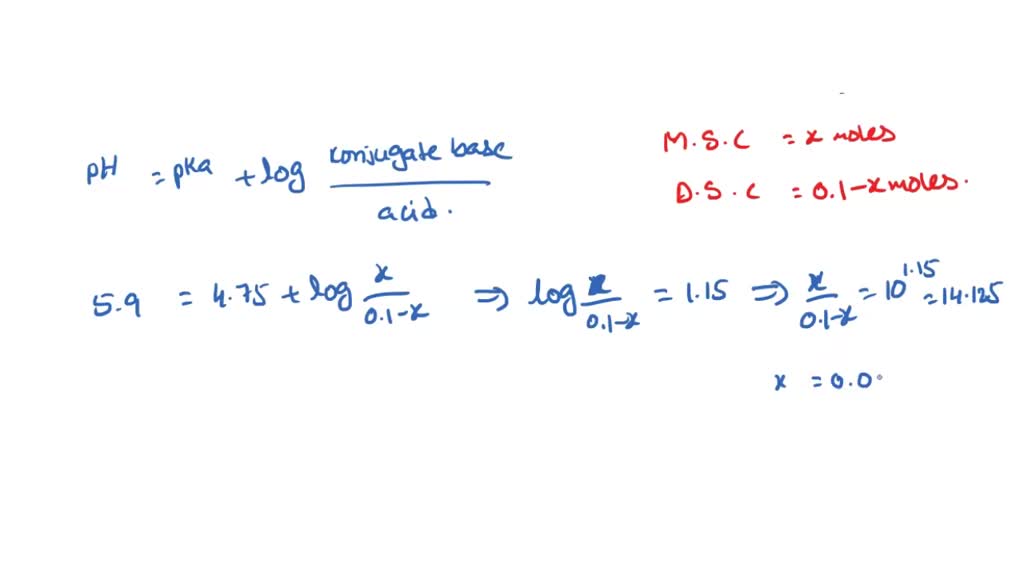





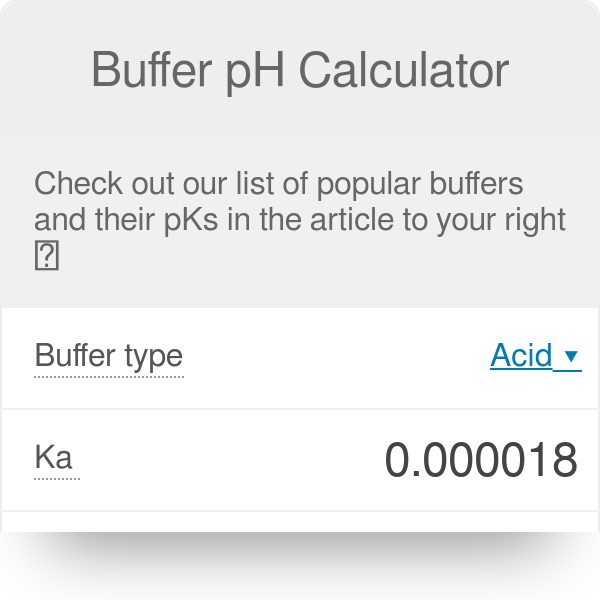

:max_bytes(150000):strip_icc()/EA4371-001-56a09bc05f9b58eba4b2083d.jpg)