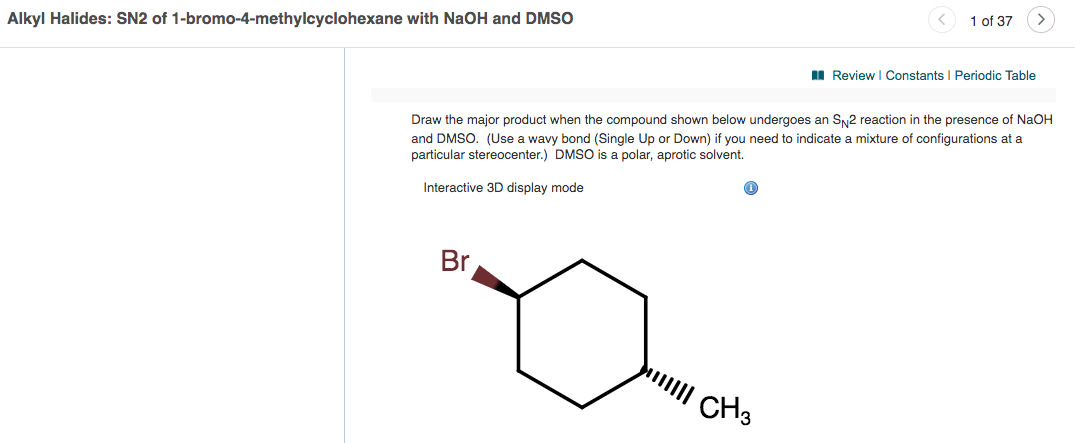
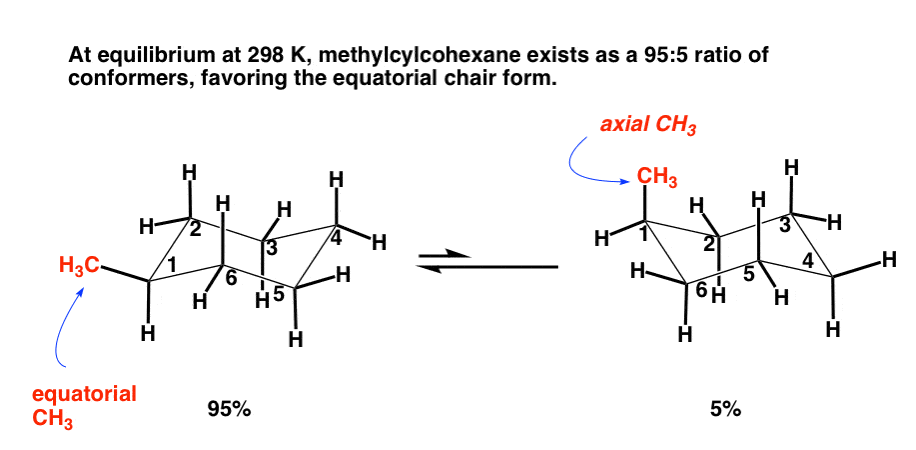
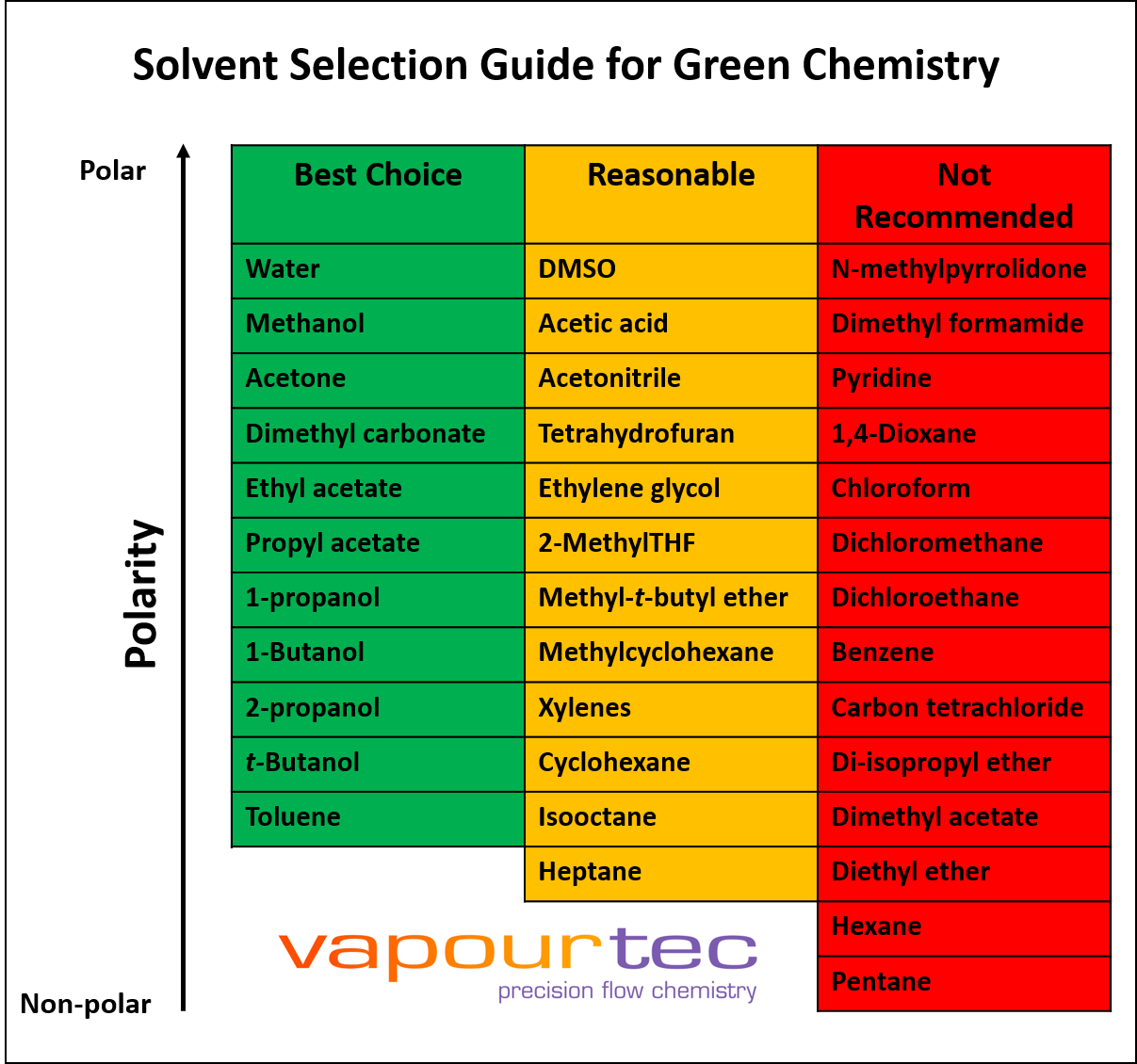
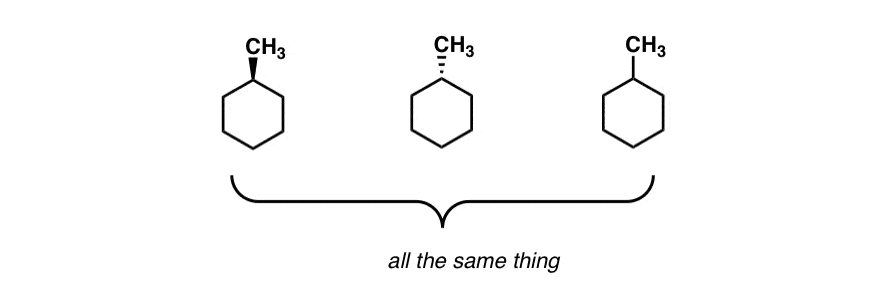
SOLVED: The solvent polarity has a substantial effect on the equilibrium between the two chair forms of trans-2-difluorocyclohexane. The following table quantifies the effect of solvent polarity on this equilibrium. Draw the

Demand for Methylcyclohexane Market Growth, Future Innovations, Recent Report 2031 | by Devidwon | Medium
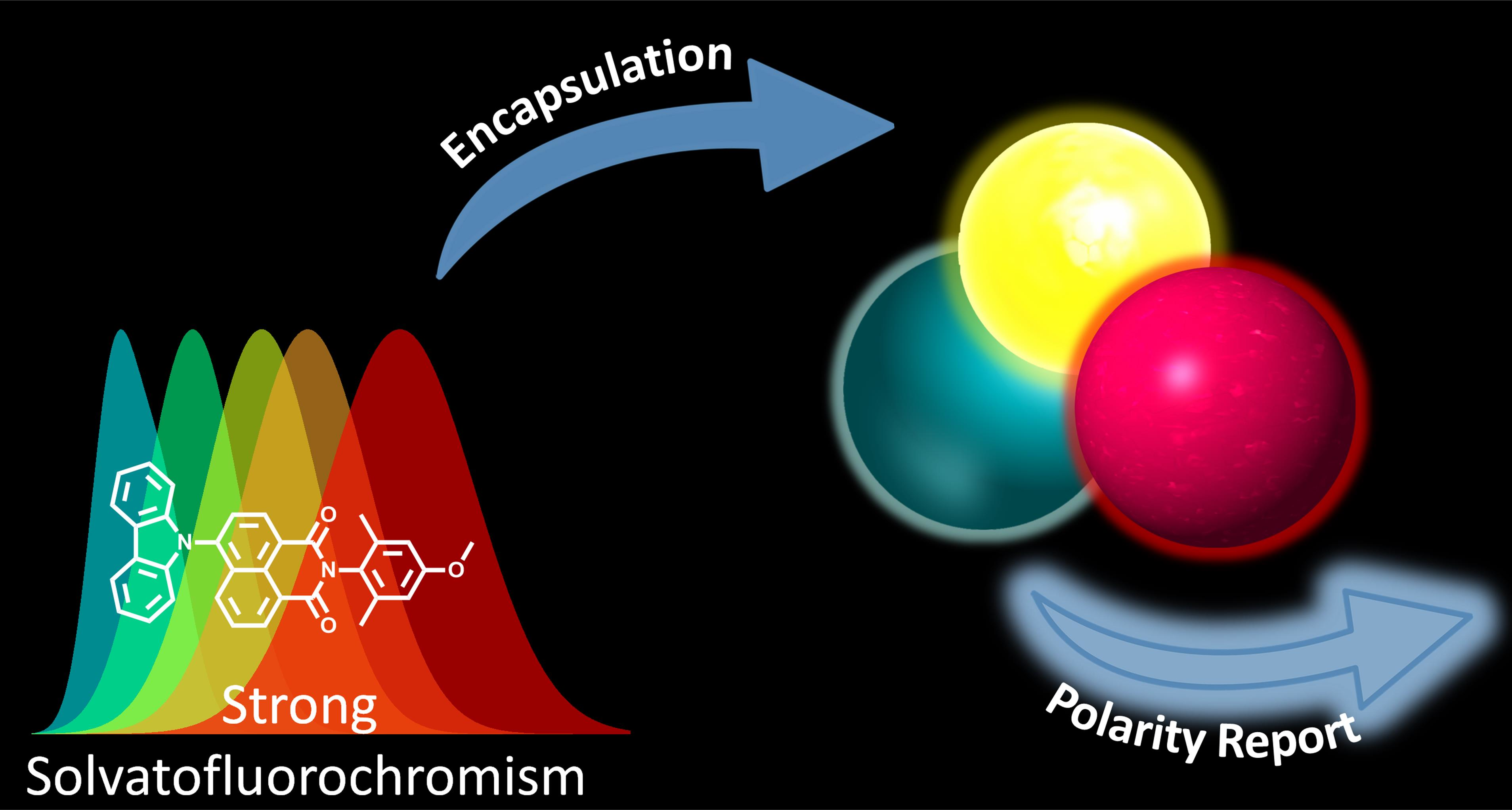
Photochem | Free Full-Text | Probing the Local Polarity in Biocompatible Nanocarriers with Solvatofluorochromism of a 4-Carbazole-1,8-naphthalimide Dye

Solvent Effect on Color, Band Shape, and Charge‐Density Distribution for Merocyanine Dyes Close to the Cyanine Limit - Würthner - 2008 - Angewandte Chemie International Edition - Wiley Online Library

Catalyst-free toluene hydrogenation to methyl-cyclohexane by pulsed DBD plasma under ambient conditions - ScienceDirect
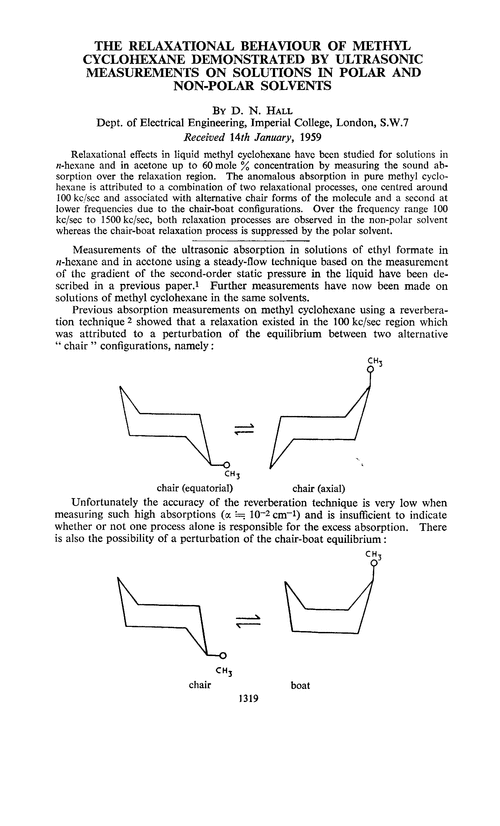
The relaxational behaviour of methyl cyclohexane demonstrated by ultrasonic measurements on solutions in polar and non-polar solvents - Transactions of the Faraday Society (RSC Publishing)
Inorganics | Free Full-Text | Study of Cyclohexane and Methylcyclohexane Functionalization Promoted by Manganese(III) Compounds

Polarity-Driven Isomerization of a Hydroxynaphthalimide-Containing Spiropyran at Room Temperature | ACS Physical Chemistry Au

For 1-bromo-3-methylcyclohexane, is the cis or trans isomer more stable? Explain. | Homework.Study.com

One of the two chair structures of cis-1-chloro-3-methylcyclohexane is more stable than the other by 15.5 \frac{kJ}{mol} (3.7 \frac{kcal}{mol}). Which is it? What is the energy cost of a 1,3-diaxial interaction between